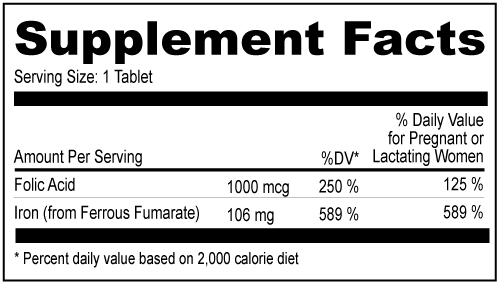
 Other Ingredients: Microcrystalline Cellulose, Sodium Starch Glycolate, Polyvinyl Alcohol, Magnesium Stearate, Polyethylene Glycol, FD & C Red 40, Talc, Hypromellose, Maltodextrin, Titanium Dioxide, FD & C Blue 2, FD & C Yellow 6 Other Ingredients: Microcrystalline Cellulose, Sodium Starch Glycolate, Polyvinyl Alcohol, Magnesium Stearate, Polyethylene Glycol, FD & C Red 40, Talc, Hypromellose, Maltodextrin, Titanium Dioxide, FD & C Blue 2, FD & C Yellow 6| NDC 52747-0306-70 | | 100 | | Tablets | | Rx only | | | NDC 52747-0306-30 | | 30 | | Tablets | | Rx only | |
| WARNING: Accidental overdose of iron-containing products is a leading cause of fatal poisoning in children under 6. Keep this product out of reach of children. In case of accidental overdose, call a doctor or poison control center immediately. | |
WARNING: Folic acid alone is improper therapy in the treatment of
pernicious anemia and other megaloblastic anemias where Vitamin B12 is
deficient.
You should contact your healthcare provider for medical advice about
adverse events. To report a serious adverse event, contact US
Pharmaceutical Corporation, P.O. Box 360465, Decatur, GA 30036.
INDICATIONS AND USAGE: For the treatment of iron
deficiency anemia and folate deficiency anemia. Hemocyte-F
is indicated in pregnancy for the prevention and treatment of
iron deficiency and to supply a maintenance dosage of folic
acid.
CONTRAINDICATIONS: Hemocyte-F is contraindicated in
patients with known hypersensitivity to any of its ingredients.
Also, all iron compounds are contraindicated in patients with
hemosiderosis, hemochromatosis, or hemolytic anemias.
Pernicious anemia is a contraindication, as folic acid may
obscure its signs and symptoms.
PRECAUTIONS: General: Anemia is a manifestation that requires appropriate investigation to determine its cause or causes. No
single regimen fits all cases, and the status of the patient observed in
follow-up is the final criterion for adequacy of therapy. Periodic clinical
and laboratory studies are considered essential. Blood examinations
including hemoglobin and hematocrit should be done at the usual
intervals to make certain therapy is adequate. Use with care in the
presence of peptic ulcer, regional enteritis and ulcerative colitis. Folic
acid, especially in doses above 0.1 mg - 0.4 mg daily, may obscure
pernicious anemia, in that hematologic remission may occur while
neurological manifestations remain progressive.
Usage in Pregnancy: Before Hemocyte-F is prescribed for
megaloblastic anemia in pregnancy, appropriate diagnostic exclusion of
Addisonian pernicious anemia, (due to faulty or blocked absorption for
Vitamin B12, or extrinsic factor, on either a genetic, immunological or
surgical basis), should be carried out.
Pediatric Use: Safety and effectiveness in pediatric patients have not
been established.
Geriatric Use: No clinical studies have been performed in patients 65
and over to determine whether older persons respond differently from
younger persons. Dosage should always begin at the low end of the
dosage scale and should consider that elderly persons may have
decreased hepatic, renal, or cardiac function, and of concomitant
disease.
ADVERSE REACTIONS: Ferrous fumarate has been implicated in
some instances of gastrointestinal disturbances, including abdominal
cramps, diarrhea, constipation, heartburn, nausea and vomiting.
Reducing the dose and administering it with meals will minimize these
effects in the sensitive patient. Increasing fiber in the diet can relieve
constipation. Iron may turn stools black. This is a harmless effect that is
a result of unabsorbed iron. Allergic sensitization has been reported
following oral administration of folic acid.
OVERDOSAGE: Iron: Signs and Symptoms: Acute overdosage of iron
may cause nausea and vomiting and, in severe cases, cardiovascular
collapse and death. Other symptoms include pallor and cyanosis,
melena, shock, drowsiness and coma. The estimated lethal dose of
orally ingested elemental iron is 300 mg per kg of body weight.
Hemocyte-F should be stored beyond the reach of children to protect
against accidental iron poisoning. Keep this product and all drugs out of
reach of children.
Treatment: For specific therapy, exchange transfusion and chelating
agents should be used. For general management, perform gastric
lavage with sodium bicarbonate solution or milk, administer intravenous
fluids and electrolytes and use oxygen.
DOSAGE AND ADMINISTRATION: Usual adult dose is one tablet
taken orally daily or as prescribed by a physician.
HOW SUPPLIED: Hemocyte-F are maroon film coated round tablets
debossed with "US" logo on one side and “F” on the other side.
Hemocyte-F are supplied in child resistant unit dose packs of 100
tablets and 30 tablets. Boxes of 100’s: NDC 52747-306-70, Boxes of
30’s: NDC 52747-306-30. Dispense in a tight, light resistant container
as defined in the USP/NF with a child resistant closure. Store at
controlled room temperature 15°C-30°C (59°F-86°F). Keep in a cool,
dry place.
Rx only.
|